1.0 Introduction
Typhoid fever is one of the most serious infectious bacterial diseases in third world countries [1]. The Salmonellae that cause significant human disease are classified in most countries under the taxon Salmonella enterica, subsp. enterica. Enteric fever caused by Salmonella enterica serotype typhi (Salmonella typhi) and Salmonella enterica serotype paratyphi A, B or C are short Gram-negative rods with rounded ends closely related to E. coli [2-3]. Enteric fever remains a major disease burden in developing countries and is associated with poor sanitation and contaminated water and food; a faecal–oral transmissible disease [4]. In areas of endemicity and in large outbreaks, most cases occur in persons aged between 3 and 29 years. Humans are the only natural host and reservoir [5].
The emergence and spread of multidrug resistant Salmonella serotypes tomany commonly used antibiotics (Ampicillin, Amoxicillin, Chloramphenicol, and trimethoprim sulfamethoxazole) is now a subject of international concern. The problem has become endemic in many developing countries, causing enormous childhood morbidity and high cost of treatment [6]. There is therefore, the need for the continuous search of indigenous plants for the treatment of typhoid fever [7]. Phytomedicines are believed to have promising potential because they contain compounds that can eliminate the quick resistance development and of low toxicity compared to synthetic drugs [3].
Piliostigma thonningii of the Fabaceae Family (subfamily Caesalpiniaceae) commonly known as camel’s foot (English), Abefe (Yoruba), Kalgo (Hausa) and Okpoatu (Igbo) is an erect perennial tree grown throughout the tropics and sub-tropics [8]. Traditionally, the plant is used in the treatment of cough, dysentery, sores, snake bites, malaria, stomach upsets and also used as a pain reliever [9]. A number of pharmacological investigations have revealed that P. thonningii has some bioactivities such as antimicrobial [10,11], antimalarial [12,13], anthelmintic [14] and antioxidant [14,15]. The present research therefore evaluates the antisalmonellal activity of the crude extract and fractions of P. thonningii and also characterize the bioactive sub-fraction using Gas Chromatography- Mass Spectrometry (GC-MS)
2.0 Materials and Methods
2.1. Collection and Identification of the plant
Matured leaves of P. thonningii were collected along Gidan kwano Road Minna, Niger State. The plant was identified at the herbarium unit of the Department of Biological Sciences, Ahmadu Bello University, Zaria where voucher number 171 was deposited.
2.2 Plant Preparation
The leaves were carefully washed under running water and air‐dried at room temperature and then milled into fine powder. About 300 grams of the powdered leaves was macerated with 1.5 liters of 70% methanol for 72 hours. The extract was filtered using a muslin cloth and subsequently evaporated using a rotatory evaporator. The semi‐dried extract was weighed, placed in sterile sample bottles and stored in a refrigerator until required for use [16].
2.3. Solvent partitioning of crude extract
The methanol crude extract was undertaken for solvent‐solvent partitioning by using the methods employed by Emran et al., [17]. The crude extract was successively partitioned by using solvents of increasing polarity in the following order; n‐hexane, chloroform and ethyl-acetate in a separating funnel. The resulting fractions of the crude extract were evaporated to dryness using rotary evaporator at 40. All the concentrated fractions were weighed and stored in air tight containers till further analysis. About one gram (1 g) of each extract and fraction were dissolved in 5 ml of 50% dimethylsulphoxide (DMSO) to make 200 mg/ml stock solution from which was serially diluted to give concentrations of 100 mg/ml, 50 mg/ml and 25 mg/ml.
2.4 Vacuum Liquid Chromatography (VLC) and Column Chromatography of Ethyl-acetate Fraction of P. thonningii
The most active partitioned fraction (ethyl-acetate fraction of P. thonningii) was further exploited in an attempt to isolate the active principle which exhibited the antibacterial activity. The method described by Amin et al., [18] was adopted for the isolation procedure. Different sub-fractions (VLC1- VLC7) were obtained by using vacuum liquid chromatography apparatus. The best fraction obtained from the VLC which exhibited significant activity against the test organisms was subjected to column chromatography using the method described by Dauda and Mudi [19] with slight modification. About 250 grams of washed silica gel (60-120 mesh size) was packed into a glass column (3.8 cm by 53 cm) in slurry of n- hexane. The fraction was dissolved in methanol and then mixed with a small quantity of silica gel, dried, triturated and then loaded on top of the column already packed with silica gel. Sequential elution was carried out using stepwise gradient solvents of increasing polarity. The process was monitored using the thin layer chromatography. An aliquot of 20 ml of the eluates were continuously collected into test tubes from the beginning to the end of the elution, in each case the eluates having similar TLC profile were pooled together into six major sub-fractions (Et1-Et6) which were further subjected to antibacterial activity.
2.5 Gas chromatography–mass spectrometry (GC-MS) analysis
The GC–MS analysis of the ethyl-acetate fraction of the Leaf extract was carried out in a (QP 2010 Plus SHIMADZU) instrument at 70 eV. One microliter (1 uL) of the extract was injected into the GC–MS using a micro syringe and the scanning was performed for 20 min.
2.6 Antisalmonellal assay
2.6.1 Test organisms
Clinical isolates of Salmonella typhi, Salmonella paratyphi A, Salmonella paratyphi B and Salmonella paratyphi C were obtained from the Microbiological laboratory of Aminu Kano teaching Hospital, Kano for the susceptibility tests. The organisms were used after their identity were confirmed at the Department of Microbiology, Bayero University, Kano. The stock culture was maintained on Nutrient agar slant at 4 in the refrigerator.
2.6.2 Antisalmonellal Susceptibility Test
The sensitivity of the crude extract and fractions were determined using the agar well diffusion method as described by Nas and Ali [20]. The prepared bacterial suspension equivalent to 0.5 McFarland Standard (1.5 x 106 CFU) was inoculated into sterile Mueller- Hinton agar medium in a sterile Petri-dish. A sterile 6 mm diameter cork borer was used to bore wells into the agar medium. The wells were filled with approximately 0.1 ml of the extract solution at a concentration of 25, 50 and100 mg/ml respectively, care was taken to prevent spillage onto the surface of the agar medium. The plates were allowed to stand on the laboratory bench for 1 hour to allow proper diffusion of the extract into the medium after which the plates were incubated at 37 ℃ for 24 hours, and thereafter the plates were observed for zones of inhibition and measured using a meter rule. The same procedure was adopted for the Column chromatography isolates. The Reference antibiotic Amoxicillin (50 mg/ml) served as control.
2.7 Statistical Analyses
The statistical analyses were carried out using statistical package for social sciences (SPSS‐ computer package). Data from the antibacterial activities of P. thonningii were expressed as mean ± standard error of three independent replicates and also subjected to one‐way analysis of variance (ANOVA) at p<0.05 level of significance for comparison of the extract activities.
3.1 Antisalmonellal activity of crude extract and fractions
The antisalmonellal activity of the crude extract and soluble fractions (n-hexane, chloroform, ethyl-acetate, aqueous methanol) are shown in Table1. The most active fraction was ethyl-acetate soluble fraction as it showed the largest zone of bacterial inhibition. The n-hexane soluble fraction showed no activity on the Salmonella species tested.
3.2 Antisalmonellal activity of VLC subfractions
The Antisalmonellal activity of all the seven subfractions obtained from Vacuum liquid chromatography of P. thonningii ethyl-acetate fractionis shown in Table 2. All the sub-fractions except sub-fraction seven (VLC7) showed zones of inhibition on at least one organism. Sub-fraction five (VLC 5) was the most active against all the Salmonella strains tested
Table 1: Zones of growth inhibition (mm) of the crude extract and fractions of the leaves of P. thonningii on the test bacteria
| Crude/ Fractions | Conc. (mg/ml) | S. typhi | S. paratyphi A | S. paratyphi B | S. paratyphi C |
| PT1 | 25 | 10.33± 0.33c | 0.00± 0.00c | 09± 0.00c | 0.00± 0.00c |
| 50 | 12.00± 0.58b | 09.33± 0.33b | 11.33± 0.33b | 9.33± 0.33b | |
| 100 | 15.33± 0.33a | 12.00± 0.58a | 14.67± 0.33a | 11.33± 0.33a | |
| PT1-01 | 25 | 0.00± 0.00a | 0.00± 0.00a | 0.00± 0.00a | 0.00± 0.00a |
| 50 | 0.00± 0.00a | 0.00± 0.00a | 0.00± 0.00a | 0.00± 0.00a | |
| 100 | 0.00± 0.00a | 0.00± 0.00a | 0.00± 0.00a | 0.00± 0.00a | |
| PT1-02 | 25 | 0.00± 0.00b | 0.00± 0.00b | 0.00± 0.00c | 0.00± 0.00c |
| 50 | 0.00± 0.00b | 0.00± 0.00b | 11.00± 0.58b | 11.67± 0.33b | |
| 100 | 11.67± 0.33a | 8.33± 0.33a | 13.33± 0.33a | 14.00± 0.00a | |
| PT1-03 | 25 | 11.33± 0.33b | 0.00± 0.00c | 12.67± 0.33c | 12.33± 0.33c |
| 50 | 11.67± 0.33b | 12.00± 0.00b | 13.67± 0.33b | 13.00± 0.58b | |
| 100 | 14.33± 0.33a | 15.33± 0.33a | 16.33± 0.33a | 15.33± 0.33a | |
| PT1-04 | 25 | 0.00± 0.00c | 0.00± 0.00c | 0.00± 0.00c | 0.00± 0.00c |
| 50 | 10.00± 0.33b | 9.33± 0.33b | 9.33± 0.33b | 9.33± 0.33b | |
| 100 | 11.00± 0.33a | 12.33± 0.33a | 11.67± 0.33a | 10.67± 0.33a |
Values are presented in means ± Standard error of three replicates. Values with the same superscript on the same column are not significantly different at p>0.05. Keys: PT1 – Methanol extract; PT1-01 – n-hexane soluble fraction; PT1-02 -Chloroform soluble fraction; PT1-03 -Ethyl-acetate soluble fraction; PT1-04 -Aqueous methanol soluble fraction
3.3 Antisalmonellal activity of Column Chromatography subfractions
Table 3 shows the results of antisalmonellal activity of subfractions obtained from Column chromatography. The Salmonella strains tested were susceptible to sub-fraction Et1 and Et2. However, Et1 indicated the strongest antibacterial activity on the test organism at low concentration of 50 mg/ml (12-15 mm).
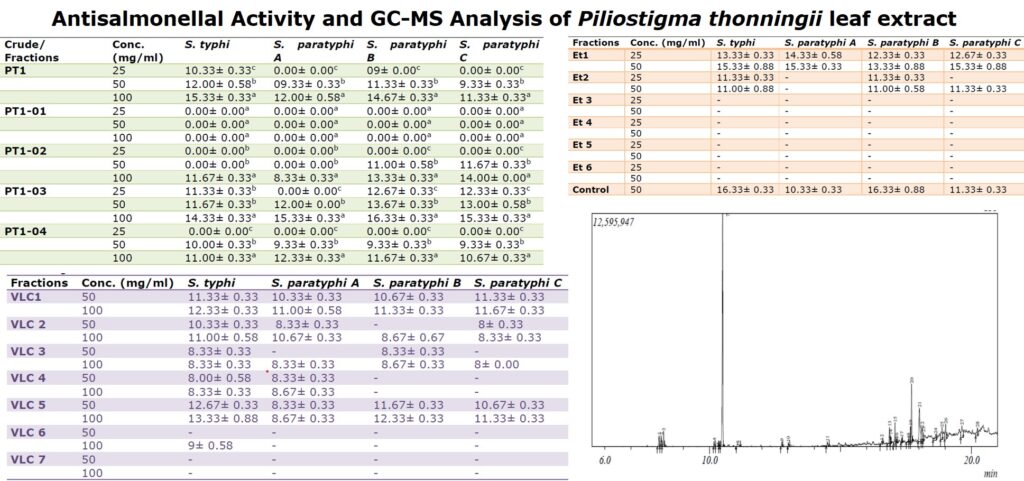
3.4 GCMS of Et1 subfraction
The chromatograph showed 28 peaks (Figure 1). The major constituents identified in the isolated compound were Levomenthol (cyclohexanol) (37.49%), n-Hexadecanoic acid (9.79%), hexadecanoic acid (Methyl ester) (6.66%) (Table 4) and many other constituents were identified as low level.
Table 2: Zones of growth inhibition (mm) of VLC subfractions obtained from ethyl-acetate fraction of P. thonningii
| Fractions | Conc. (mg/ml) | S. typhi | S. paratyphi A | S. paratyphi B | S. paratyphi C |
| VLC1 | 50 | 11.33± 0.33 | 10.33± 0.33 | 10.67± 0.33 | 11.33± 0.33 |
| 100 | 12.33± 0.33 | 11.00± 0.58 | 11.33± 0.33 | 11.67± 0.33 | |
| VLC 2 | 50 | 10.33± 0.33 | 8.33± 0.33 | – | 8± 0.33 |
| 100 | 11.00± 0.58 | 10.67± 0.33 | 8.67± 0.67 | 8.33± 0.33 | |
| VLC 3 | 50 | 8.33± 0.33 | – | 8.33± 0.33 | – |
| 100 | 8.33± 0.33 | 8.33± 0.33 | 8.67± 0.33 | 8± 0.00 | |
| VLC 4 | 50 | 8.00± 0.58 | 8.33± 0.33 | – | – |
| 100 | 8.33± 0.33 | 8.67± 0.33 | – | – | |
| VLC 5 | 50 | 12.67± 0.33 | 8.33± 0.33 | 11.67± 0.33 | 10.67± 0.33 |
| 100 | 13.33± 0.88 | 8.67± 0.33 | 12.33± 0.33 | 11.33± 0.33 | |
| VLC 6 | 50 | – | – | – | – |
| 100 | 9± 0.58 | – | – | – | |
| VLC 7 | 50 | – | – | – | – |
| 100 | – | – | – | – |
Key: – No zone of inhibition
Table 3: Zones of growth inhibition (mm) of isolates obtained from Column Chromatography isolates of P. thonningii
| Fractions | Conc. (mg/ml) | S. typhi | S. paratyphi A | S. paratyphi B | S. paratyphi C |
| Et1 | 25 | 13.33± 0.33 | 14.33± 0.58 | 12.33± 0.33 | 12.67± 0.33 |
| 50 | 15.33± 0.88 | 15.33± 0.33 | 13.33± 0.88 | 15.33± 0.88 | |
| Et2 | 25 | 11.33± 0.33 | – | 11.33± 0.33 | – |
| 50 | 11.00± 0.88 | – | 11.00± 0.58 | 11.33± 0.33 | |
| Et 3 | 25 | – | – | – | – |
| 50 | – | – | – | – | |
| Et 4 | 25 | – | – | – | – |
| 50 | – | – | – | – | |
| Et 5 | 25 | – | – | – | – |
| 50 | – | – | – | – | |
| Et 6 | 25 | – | – | – | – |
| 50 | – | – | – | – | |
| Control | 50 | 16.33± 0.33 | 10.33± 0.33 | 16.33± 0.88 | 11.33± 0.33 |
Key: – No zone of inhibition
3.4 GCMS of Et1 subfraction
The chromatograph showed 28 peaks (Figure 1). The major constituents identified in the isolated compound were Levomenthol (cyclohexanol) (37.49%), n-Hexadecanoic acid (9.79%), hexadecanoic acid (Methyl ester) (6.66%)(Table 4) and many other constituents were identified as low level.
Table 4: The major chemical constituents of Column isolate Et1 by GC-MS
| S/N | Peak | Retention time | Peak % | Derivatives | Molecular Formular |
| 1 | 7 | 10.50 | 37.49 | Levomenthol | C10H20O |
| 2 | 20 | 17.70 | 6.66 | Hexadecanoic acid | C17H34O2 |
| 3 | 21 | 18.00 | 9.79 | n-Hexadecanoic acid | C16H32O2 |
4.0 Discussion
Plant extracts are considered to be valuable source of biological active compounds. In this study the antibacterial activity of the crude extracts and fractions were assessed against some Salmonella strains. The crude extract andall the other soluble fractions (n-hexane, chloroform, ethyl-acetate, aqueous methanol) had antibacterial activity against all the tested organisms except n-hexane soluble fraction. This could be that the bioactive compounds present in the Plant were more soluble in the polar solvents as compared to the non‐polar solvent. Different extracts from same plant can show different constituents and antimicrobial activities on the same organism [19]. However, the most active fraction was ethyl-acetate soluble fraction as it showed the largest zone of bacterial inhibition as such was subjected to Vacuum liquid chromatography.
All the subfractions except sub-fraction seven (VLC7) showed zones of inhibition on at least one organism. Fraction five (VLC 5) was the most active against the bacteria strains tested, as a result of which, it was further purified using column chromatography. The Salmonella strains tested were susceptible to the column chromatography isolate Et1 and Et2. However, Et1 indicated the strongest antibacterial activity on the test organism at low concentration of 50 mg/ml (12-15 mm) when compared with the crude extract and subfractions. The activities of the isolates were significant as compared to that of the reference antibiotic Amoxicilin (control) used in the study. The present study has shown that antibacterial activity varies with the fractions. The observed difference could be attributed to the variation in the distribution of active principles according to their affinity for the solvent used in fractionation [21].
Few investigations on the antibacterial properties of P. thonningii have been reported. Ewansiha et al. [22] and Chukwunonye et al.[11] had also reported the potency of the leaf extract and fractions of P. thonningii on S. typhi and other bacteria. Gas chromatography–mass spectrometry (GC-MS) is an analytical method that combines the features of gas-chromatography and mass spectrometry to identify different substances within a test sample. Gas chromatography has gained widespread acceptance in numerous application areas, such as process control in chemical plants, quality control in the food industry, monitoring sample composition in the oil-industry, environmental and bio medical sciences [23]. The interpretation on mass spectrum was conducted using the database of National Institute Standard and Technology (NIST) having more than 62,000 patterns. The spectrum of the unknown component was compared with the spectrum of the known components stored in the NIST library.
Twenty-eight compounds were identified inthe column isolate by GC-MS analysis. The compounds with the highest composition percentage were cyclohexanol (Levomenthol), n-hexadecanoic acid (palmitic acid) and hexadecanoic acid. The identified compounds possess many biological properties. Menthol possesses antibacterial and antioxidant activity [24]. n-Hexadecanoic acid and hexadecanoic acid have also been reported as major constituents in the GCMS analysis of some medicinal plants [23, 25-26], these compounds acts as an antimicrobial, antioxidant, hypocholesterolemic, nematicide, pesticide and lubricant activities [27-29]. The presence of the various bioactive compounds identified in this plant justifies its use as medicine for various ailments by traditional practitioners.
4.0 Conclusion
The antisalmonellal activity of P. thonningii leaf extract showed that the extracts and fractions were effective against the Salmonella strains with the column chromatography isolate exhibiting the highest activity when compared to the other fractions. The antibacterial activity of the plant may be due to the presence of the bioactive compounds identified in this study using gas chromatography–mass spectrometry. The ethyl-acetate fractions and the identified column isolates may therefore be a readily available source of cheap and potent antibacterial agents to be used in the therapy of infections caused by these often multi resistant organisms. These findings provide a rationale for the use of the plant in traditional medicine.
Acknowledgement: This study was part of a research project funded by the TETFUND Institution based research intervention (IBRI) Fund (TETFUND/FUTMINNA/2016-2017/6th BRP/18) Federal University of Technology Minna, Niger State, Nigeria.
Authors Contributions: The work was conducted in collaboration of all authors. All authors read and approved the final version of the manuscript
Conflicts of interest: The authors declare that they have no competing interests.
References
- Banerji, P., Banerji, P., Chandra, D.G., Aminul, I.,Kumar, M.S and Mukhopadhyay, Susmita (2012) “Efficacy of Baptisia tinctoria in the Treatment of Typhoid: Its Possible Role in Inducing Antibody Formation. Journal of Complementary and Integrative Medicine, 9(1), 1-13
- Kayser, F.H., Kurt, A.B., Eckert, J. and Zinkernage, R.N. (2005). Medical Microbiology, 5th ed. Thieme, New York, USA, p. 362, Appl, Wemding.
- Ruparelia, J., Rabari, A., Joshi, N., Dhediya, A., Halpati, J., Patel, A. and Jha, C.K. (2021). Reconnoitring natural Antibacterial appraisal of Medicinal Plants extract against Human pathogen Salmonella paratyphi A and Salmonella paratyphi B. Chemical and Biomolecular Engineering, 6(2), 30-36.
- Buckle, G. C., Walker, C. L. and Black, R. E. (2012). Typhoid Fever and Paratyphoid Fever: Systematic Review to Estimate Global Morbidity and Mortality For 2010. Journal of Global Health, 2, 1-4.
- Tadesse, G. (2014). Prevalence of Human Salmonellosis in Ethiopia: a systematic review and meta-analysis. BMC Infectious Diseases, 14, 88-98.
- Bekoe, E.O., Agyare, C., Sarkodie, J & Dadebo, D. (2017). Herbal Medicines used in the treatment of Typhoid in the Ga East Municipality of Ghana. International journal of tropical disease and Health, 23(4), 1-13
- Sarkiyayi S, and Abdulrasheed K. (2013). Preliminary Investigation on Anti Typhoid properties of Acacia nilotica Leaf extract. Global J Med Res, 13 (5), 10-15
- Jimoh, F.O. and A.T. Oladeji, (2005). Preliminary Studies of Piliostigma thonningii seeds: Proximate analysis, mineral Composition and Phytochemical screening. African Journal of Biotechnology, 4(12), 1439-1442.
- Egharevba, H.O. and F.O. Kunle, (2010). Preliminary Phytochemical and Proximate Analysis of the Leaves of Piliostigma thonningii (Schumach.) Milne-Redhead. Ethnobotanical Leaflets, 14, 570-577
- Daniyan, S.Y., Galadima, M, Ijah U.J.J, Odama L.E., Yusuf, A.Y., Jigam, A.A. and Abbas, Y. (2011). Evaluation of Antibacterial activity of Piliostigma thonningii (Crude Extract) and Fractions 1-3, 4, 5, 6 And 7 against Methicillin-Resistant Staphylococcus aureus (MRSA). Research Journal of Pharmaceutical, Biological and Chemical Sciences,1 (4), 173.
- Chukwunonye, U.C.E., Ebele, O.P., Kenne, T.M. and Gaza, A.S.P. (2017). Phytochemical Screening and Antimicrobial Activity of Methanol Extract and Fractions of the Leaf of Piliostigma thonningii Schum (Caesalpiniaceae). World Applied Sciences Journal, 35(4), 621-625
- Kwaji, A., Bassi, P.U., Nneji, C.M, and Ademowo, G. (2010). Preliminary studies on Piliostigma thonningii Schum leaf extract: Phytochemical screening and in vitro antimalarial activity. African Journal of Microbiological Research,4(9),735- 739.
- Madara, A.A., Ajayi, J.A., Salawu, O.A., and Tijani, A.Y. (2010). Anti-malarial activity of ethanolic leaf extract of Piliostigma thonningii Schum. (Caesalpiniaceae) in mice infected with Plasmodium berghei berghei. African Journal of Biotechnology, 9(23), 3475-3480
- Koma, O.S and Rufai, Y. (2015). Antimicrobial and Antioxidant Potentials, and chemical constituents of the Leaf extracts of the Nigerian Piliostigma thonningii (Caesalpiniaceae) Schum. European Journal of Medicinal Plants, 7(3), 137-145
- Taofeek, O.A. (2011). In vivo Antioxidant potentials of Piliostigma thonningii (Schum) leaves: Studies on hepatic marker enzyme, antioxidant system, drug detoxifying enzyme and lipid peroxidation. Human Experimental Toxicology, 30(1), 55- 62.
- Tiwari, P., Kumar, B., Kaur, M., Kaur, G., and Kaur, H. (2011). Phytochemical screening and Extraction: A Review. Internationale Pharmaceutica Sciencia, 1(1), 98-106
- Emran, T., Rahman A, Nasiruddin, M.N., Rahman, M., Uddin, Z., Dash, R & Layzu C. (2015). Effects of organic extracts and their different fractions of five Bangladeshi plants on in vitro thrombolysis. BMC Complementary and Alternative Medicine, 15, 128‐135.
- Amin, N., Qadir, M.I., Khan, T.J., Abbas, G., Ahmad, B., Janbaz, K.H. and Ali, M. (2012). Antibacterial Activity of Vacuum Liquid Chromatography (VLC) Isolated Fractions of Chloroform Extracts of Seeds of Achyranthes aspera. Journal Chemical Society of Pakistan, 34(3), 589-592
- Dauda, U. And Mudi, S.Y. (2013). Screening and Bioassay-Guided Isolation of antimicrobial Components from Laggera mollis. Bayero Journal of Pure and Applied Sciences,6(1),152 – 158
- Nas, F. S. & Ali, M. (2017). Antibacterial Activity of Boswellia dalzielii Leaves Extracts against Some Pathogenic Bacterial Isolates. Journal of Advances in Microbiology, 7(1), 1-8
- Jamil, M., Haq,I., Mirza, B. and Qayyum, M. (2012). Isolation of antibacterial compounds from Quercus dilatata L. through bioassay guided fractionation. Annals of Clinical Microbiology and Antimicrobials,11,1-11
- Ewansiha, J.U., Okafor, A.C, Doughari, J, Busari, M.B. (2015). Antibacterial activity of the leaf extract of Piliostigma thonningii against Salmonella typhi and Shigella dysenteriae. Adv Med Plant Res, 3(4), 151-154.
- Chukwunonye, U.C.E., Ebele, O.P., Kenne, T.M. and Gaza, A.S.P. (2017). Phytochemical Screening and Antimicrobial Activity of Methanol Extract and Fractions of the Leaf of Piliostigma thonningii Schum (Caesalpiniaceae). World Applied Sciences Journal, 35 (4), 621-625
- Sermakkani, M and Thangapandian, V. (2012). Gc-Ms Analysis of Cassia italica Leaf Methanol extract. Asian J Pharm Clin Res, 5(2),90-94
- Fagbemi, K.O., Aina, D.A., Coopoosamy, R.M. & Olajuyigbe, O.O., (2022). Gas chromatography-mass spectrometry chemical profile investigation and biological activities of ethylacetate fraction of Baobab (Adansonia digitata L.) pulp used in the treatment of urinary tract infections, Journal of Medicinal Plants for Economic Development, 6(1), 1-10
- Rajeswari, G., Murugan, M. & Mohan, V. R. (2013). GC-MS analysis of Bioactive components of Hugonia mystax L. (Linaceae). Research Journal of Pharmaceutical, Biological and Chemical Sciences, 3 (4), 301-308.
- Adefolalu, F.S., Ogbadoyi E.O., Ndams, I.S & Mann, A. (2015). Larvicidal Activities of N-Hexane Fraction of Ocimum gratissimum Leaf against Mosquito Larvae and its GC-Ms Analysis of Phytoconstituents, Journal of Applied Life Sciences International, 2(4), 175-188,
- Shibula, K & Velavan, S. (2015). Determination of Phytocomponents in Methanolic Extract of Annona muricata Leaf Using GC-MS Technique. International Journal of Pharmacognosy and Phytochemical Research, 7(6), 1251-1255
- Gobalakrishnan, R., Manikandan, P. and Bhuvaneswari, R. (2014). Antimicrobial potential and Bioactive constituents from aerial parts of Vitis setosa wall. Journal of Medicinal Plant Research, 8 (11), 454-460