- Introduction
The increase in price of conventional feed ingredients for animals has led researchers to work on alternative feed source which are non-conventional and less competed for between animals and humans (especially plants and animal by-products, kitchen wastes and left-over) to reduce the cost of production [1].
Feeding of livestock with these by-products comes with its limitations because some of them contains some nutrients which when consumed in excess can result into physiological problems which when treated with conventional commercial medications results to increase in cost of production [2].
Rabbit, as a micro-livestock, is an economic animal that could bridge the wide gap in dietary protein requirements in Nigeria because it is socially acceptable, requires less space for rearing and absence of religious taboo on its rearing and consumption as well as peculiar digestive physiology which permits the use of forages and agro-industrial by-products thus making it a less competitive species with man for cereal and legume grain [2]. In addition, rabbits are efficient converters of feed to meat and can utilize up to 30% fibre as against 10% fibre utilized by most poultry species [3]. Although rabbits can survive on all forage diets, but optimum performance can only be insured in a mixed feeding regime involving forage and formulated feed [4].
Survival of these animal species is highly dependent on their physiological status and diets which at times often leads to health issues like Atherosclerosis, (a major cardiovascular disease (CVD) in man and animal which can shorten the life span). Apart from mortality that may be recorded, the cost of medication will also lead to increase in the cost of production [5]. While the use of orthodox lipid lowering agents is expensive and associated with side effects like flatulence, constipation, and dyspepsia [4], the screening of herbs used in the traditional medicine in the management of CVD for lipid lowering effect has gained wide scientific interest in the last decades [5].
Garcinia kola which belongs to the family of Guttiferae also known asꞌꞌBitter kolaꞌꞌ in English are known to have hypolipidemic effects on serum lipids, thus it can serve the lipid the lipid lowering purpose in the serum. The seed is commonly found in the West African countries and its natural habitat is subtropical or tropical moist low land forests [1]. The growing tree and roots of Garcinia kola provide chew-sticks and the seeds are eaten raw [6]. The extract of the seed has been proven scientifically to have several pharmacological activities which include anti-inflammatory, analgesic, molluscidal, anti-atherogenic, antioxidant and hepato-protective activities [6,7,8,9]. The present work was aimed at evaluating the effect of fortifying the feed of rabbits with graded level of processed G. kola seed on the serum lipid profile of weaner rabbits fed lipid rich diets.
2.0 Materials and Methods
2.1 Study Location
The study was conducted at the small animal experimental Unit of the Department of Animal Health and Production Technology, Federal College of Wildlife Management, New-Bussa, Niger State, Nigeria between the months of February and May 2022.New- Bussa lies on latitude 90 52ꞌ 59.99ꞌꞌ N and Longitude 40 30ꞌ 59.99ꞌꞌ E.
2.2 Plant Material
The freshly harvested fruits of G. kola (Bitter kola) were bought from Wawa market, Borgu Local Government Area of Niger State, Nigeria in the month of January 2022 and were authenticated in the herbarium of Nigeria Institute of Science Laboratory Technology (NISLT), Samonda, University of Ibadan Road, Ibadan, Oyo State, Nigeria with Laboratory number 20121164 – 20121165 and Code number460/2021.
2.3 Sample Preparation
The outer testa of each G. kola seed was removed washed and air dried for about twenty-four (24) hours. Each seed was cut into smaller pieces and the resulting pellets were dried in hot air oven (Gallenkamp) for about twelve (12) hours at 40ºC as described by [5] and the dry seed pellets were ground to fine powder. The resulting powder was stored in an airtight container throughout the experimental period.
2.4 Experimental Animals
Forty (40) unsexed New Zealand white breed of Weaner rabbits weighing 0.90 ± 0.02Kg used for this experiment were sourced from the small animal unit of the department of Animal Health and Production Technology, Federal College of Wildlife Management, Forestry Research Institute, New-Bussa in Niger State. They were fed with the compounded and pelleted (to prevent aspiration pneumonia) experimental diet ad–libitum in well-ventilated cages as shown in table 1. They were supplied with clean drinking water throughout the experimental period. Ethical conditions governing the conduct of experiments with live animals were strictly observed as recommended by [10].
2.5 Experimental Design
The experimental animals were randomly divided into five treatments containing eight rabbits per treatment and each treatment replicated four times with two rabbits per replicate in a Completely Randomized Design (CRD) and were labeled T1, T2, T3, T4 and T5 respectively. All the rabbits were fed on the same diet which is lipid rich with a very high metabolizable energy (M/E). T1 served as the control and received plain feed without seed sample. Treatments 2 to5 were fed feeds fortified with graded level of the processed G. kolaseeds at 5g/100g, 10g/100g, 15g/100g and 20g/100grespectively. The animals were fed at 10% of their body biomass for fifty-six (56) days.
2.6 Proximate Analysis of the Experimental Diets
Standard methods of the Association of Official Analytical Chemists [3] were used to determine the moisture, crude protein (CP), ether extract (EE), ash, nitrogen free extract (NFE) and crude fiber (CF) contents of the experimental feed samples as described by [5]. Each analysis was carried out in quadruplets and the result is as shown on table 1.
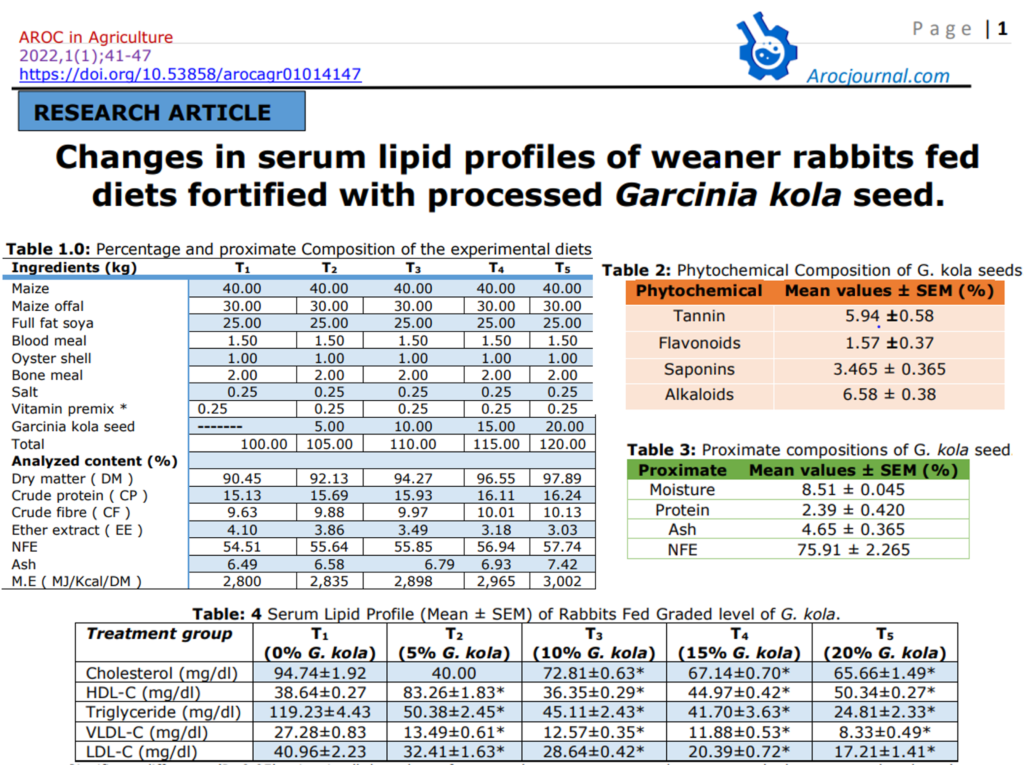
Table 1.0: Percentage and proximate Composition of the experimental diets
| Ingredients (kg) | T1 | T2 | T3 | T4 | T5 |
| Maize | 40.00 | 40.00 | 40.00 | 40.00 | 40.00 |
| Maize offal | 30.00 | 30.00 | 30.00 | 30.00 | 30.00 |
| Full fat soya | 25.00 | 25.00 | 25.00 | 25.00 | 25.00 |
| Blood meal | 1.50 | 1.50 | 1.50 | 1.50 | 1.50 |
| Oyster shell | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 |
| Bone meal | 2.00 | 2.00 | 2.00 | 2.00 | 2.00 |
| Salt | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 |
| Vitamin premix * | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 |
| Garcinia kola seed | ——- | 5.00 | 10.00 | 15.00 | 20.00 |
| Total | 100.00 | 105.00 | 110.00 | 115.00 | 120.00 |
| Analyzed content (%) | |||||
| Dry matter ( DM ) | 90.45 | 92.13 | 94.27 | 96.55 | 97.89 |
| Crude protein ( CP ) | 15.13 | 15.69 | 15.93 | 16.11 | 16.24 |
| Crude fibre ( CF ) | 9.63 | 9.88 | 9.97 | 10.01 | 10.13 |
| Ether extract ( EE ) | 4.10 | 3.86 | 3.49 | 3.18 | 3.03 |
| NFE | 54.51 | 55.64 | 55.85 | 56.94 | 57.74 |
| Ash | 6.49 | 6.58 | 6.79 | 6.93 | 7.42 |
| M.E ( MJ/Kcal/DM ) | 2,800 | 2,835 | 2,898 | 2,965 | 3,002 |
*Vitamin premix is to provide the following per kg diet: Vit, A, 1500IU; Vit E, 11.0mg; Riboflavin, 9.0mg; Biotin, 0.25; Pantothenic acid, 11.0mg; Vit k , 3.0mg; B1 , 2.5mg; B2, 0.3mg; B6 , 8.0mg; Nicotinic acid, 8.0 mg; Fe, 5.0mg; Mn, 10.0mg; Zn, 4.5mg; Co,0.2mg; Se,0.01mg.**NFE: Nitrogen free extract**ME: Metabolizable Energy.
2.7 Sample Collection
Twenty-four (24) hours after the last feed administration, blood sample for sera preparation were collected by direct cardiac puncture into sterile plain sample bottles. The serum samples were separated from the clot by centrifugation at 3000rpm for 5min using bench top centrifuge (MSE minor, England®). Serum samples were harvested into sterile plain sample bottles and stored in refrigerator for serum lipid profile analysis.
2.8 Biochemical Analysis
The serum lipid profiles of the rabbits were evaluated using a commercially available assay kit (Randox, UK) as described by [5]. The serum level of total cholesterol (TC) was measured by enzymatic hydrolysis and oxidation method as described by [11]. The serum triglyceride (ST) level was determined after enzymatic hydrolysis of the sample with lipases as described by [12]. The serum level of High Density Lipoprotein Cholesterol (HDL-C) was measured by the method of [13]. The serum Very Low-Density Lipoprotein Cholesterol (VLDLC)was calculated as 1/5 of the ST [14], while the serum Low Density Lipoprotein Cholesterol (LDLC) was calculated using the formula described by [15].
2.9 Photochemical Analysis of G. kola
Tannins, Saponins, Flavonoids and Total alkaloids of the seed samples were determined using the method described by [16]and [17] and the result is as shown on table 2.
2.9.1 Determination of Alkaloids
Five (5) grams of the plant sample was placed in a 250ml beaker and 200ml of 10% ethanoic acid in ethanol was added. The mixture was covered and allowed to stand for 4 hours. It was then filtered and the filtrate was concentrated on a water bath until it reaches a quarter of its original volume. Concentrated ammonium hydroxide was added until precipitation was complete. The mixture was allowed to settle and the precipitate collected on a weighed filter paper and washed with dilute ammonium hydroxide. The precipitate, alkaloid, was dried and weighed. The percentage alkaloid was calculated by difference [5].
2.9.2 Determination of Flavonoids
Ten (10) grams of plant sample was repeatedly extracted with 100ml of 80% aqueous methanol at room temperature. The mixture was then filtered through a filter paper into a pre-weighed 250ml beaker. The filtrate was transferred into a water bath and allowed to evaporate to dryness and weighed. The percentage flavonoid was calculated by difference [5].
2.9.3 Determination of Saponins
Twenty (20) grams of seed sample was weighed into a 250ml conical flask. One hundred milliliters of 20% ethanol was added. The mixture was heated over a hot water bath for 4 hours with continuous stirring at about 55ºC. It was then filtered with a Whatman No.1 paper. The residue was re-extracted with another 200ml of 20% ethanol. The combined extract was reduced to 40ml over a water bath at about 90ºC. The concentrated extract was then transferred into a 250ml separator funnel and 20ml of diethyl ether was added to the extract and shaken vigorously. The aqueous layer was recovered while the diethyl ether layer was discarded. This purification process was repeated. Sixty (60) ml of n-butanol was added and the combined n-butanol extract was washed twice with 10ml of 5% sodium chloride. The remaining solution was then heated on a water-bath in a pre-weighed 250ml beaker. After evaporation the residue was dried in a Gallenkamp moisture extraction oven (Size 1) to a constant weight. The % Saponin was calculated by difference [5].
2.9.4 Tannin determination
The seed sample, 500mg, was weighed into a 50ml plastic bottle. 50ml of distilled water was added and shaken for 1h in a mechanical shaker. This was filtered into a 50ml volumetric flask and made up to the mark. Then 5ml of the filterate was pipetted out into a test tube and mixed with 2ml of 0.1M ferric chloride in 0.IN hydrochloric acid and 0.008M potassium ferrocyanide. The absorbance was measured at 700nanometers within 10minutes [5].
2.9.5 Proximate Analysis of Garcinia kola
The values of moisture content, crude protein (CP), Ether extracts (EE), Nitrogen Free Extracts (NFE) and Crude fibre of the seed samples were determined by using the Standard methods described by the Association of Official Analytical Chemists [18].
2.9.6 Determination of the Moisture content
Moisture content was determined by heating 2.0g of each fresh sample to a constant weight in a crucible placed in an oven maintained at 105ºC. The dry matter was used in the determination of the other parameters.
2.9.7 Determination of the Crude Protein (CP)
Crude protein (% total nitrogen X 6.25) was determined by the Kjeldahl method, using 2.0g samples. Ether extract was obtained by exhaustively extracting 5.0g of each sample in a Soxhlet apparatus using petroleum ether (boiling point range 40o-60ºC) as the extractant.
2.9.8 Ash content determination
Ash was determined by the incineration of 2.0g sample placed in a muffle furnace maintained at 550ºC for 5h. Crude fiber was obtained by digesting 2.0g of sample with 1.25% w/v Sulphuric acid and 1.25% w/v sodium hydroxide, the fiber was filtered, dried at 120ºC, weighed and incinerating the residue in a muffle furnace maintained at 550ºC for five hours (5hrs).
2.9.9 Determination of Nitrogen Free Extracts (NFE)
Nitrogen free extracts (NFE) which represents soluble carbohydrates and other digestible and easily utilizable non-nitrogenous substances in feed were determined by mathematical calculation. It was obtained by subtracting the sum of the percentages of all the nutrients already determined from 100 by using the formula below;
%NFE=100-(%moisture+%CF+%CP+%EE+%Ash)
2.9.10 Statistical Analysis
Data obtained were analyzed using one-way analysis of variance (ANOVA) and the variant means were separated by least significant difference (LSD) of the different groups. Significance (p<0.05) was accepted at 5% level.
3.0 Results and Discussion
3.1 Phytochemical Composition
The result of the phytochemical analysis obtained for the seed sample of G. kola in this research work is not in agreement with the reports of [19] who reported the values of 0.342% for Tannins, 2.471% for Saponin, 0.645% for Alkaloids and 2.041% for flavonoids, but the results are in agreement with the works of [5], that reported the values of 5.08 ± 0.2% for Tannin, 0.93± 0.03% for Flavonoids, 5.13 ± 0.67% for Alkaloids and 2.54±0.01% for saponin. The difference in the values might be due to the fact that the seeds were sourced from different locations and they grew in different environment.
Table 2: Phytochemical Composition of G. kola seeds.
| Phytochemical | Mean values ± SEM (%) |
| Tannin | 5.94 ±0.58 |
| Flavonoids | 1.57 ±0.37 |
| Saponins | 3.465 ± 0.365 |
| Alkaloids | 6.58 ± 0.38 |
3.2 Proximate Composition
Results of proximate nutrient composition of G. kolaon table 3shows that the values of the moisture content, Crude protein, Ash content and NFE varies between 8.51 ± 0.045, 2.39 ± 0.420, 4.65 ± 0.365 and 75.91 ± 2.26 respectively. This result is not in agreement with the works of [9] who reported that G. kola seed contained 97.31, 39.52, 43.25, 11.40, 114.02 and 694.48 g/kg dry weight of moisture, CP, EE, ash, CF and NFE respectively. Also, [5] reported that G. kola seed contained 60.48, 2.48, 4.51, 0.79, 5.23 and 35.64% of moisture, CP, EE, Ash, CF and NFE respectively. These differences might not be unconnected with the source of the materials used [5].
Table 3: Proximate compositions of G. kola seed.
| Proximate | Mean values ± SEM (%) |
| Moisture | 8.51 ± 0.045 |
| Protein | 2.39 ± 0.420 |
| Ash | 4.65 ± 0.365 |
| NFE | 75.91 ± 2.265 |
3.3 Serum lipid profile
The results of this study shown on table 5 revealed that feeding of the New-Zealand breed of weaner rabbits with feed fortified with powdered G. kola seed produced a significant (p<0.05) dose-dependent serum-lipid lowering effects which may be mediated by some of its phytochemical and nutritional constituents, this in agreement with the works of [20] who fortified the feed of Albino Winstar Rat with G. kola and obtained similar results. Though the mechanism of lipid lowering effect is not known, it could be either through reduction in absorption of cholesterol from the gut or by reduction in the biosynthesis of cholesterol [5].
The possible mechanism of reducing dietary cholesterol absorption from the gut is by binding of the phytochemical constituent such as Phytosterols to the cholesterol receptor site in the gut mucosa [21].
Table: 4 Serum Lipid Profile (Mean ± SEM) of Rabbits Fed Graded level of G. kola.
| Treatment group | T1 (0% G. kola) | T2 (5% G. kola) | T3 (10% G. kola) | T4 (15% G. kola) | T5 (20% G. kola) |
| Cholesterol (mg/dl) | 94.74±1.92 | 40.00 | 72.81±0.63* | 67.14±0.70* | 65.66±1.49* |
| HDL-C (mg/dl) | 38.64±0.27 | 83.26±1.83* | 36.35±0.29* | 44.97±0.42* | 50.34±0.27* |
| Triglyceride (mg/dl) | 119.23±4.43 | 50.38±2.45* | 45.11±2.43* | 41.70±3.63* | 24.81±2.33* |
| VLDL-C (mg/dl) | 27.28±0.83 | 13.49±0.61* | 12.57±0.35* | 11.88±0.53* | 8.33±0.49* |
| LDL-C (mg/dl) | 40.96±2.23 | 32.41±1.63* | 28.64±0.42* | 20.39±0.72* | 17.21±1.41* |
*Significant difference (P<0.05) exists in all the values of measured parameters across the treatments) when compared to the values obtained from the negative control…HDL-C= High Density Lipoprotein Cholesterol, VLDL-C= Very Low-Density Lipoprotein Cholesterol, LDL-C= Low Density Lipoprotein Cholesterol.
Garcinia kola has been shown to possess several pharmacological activities such as; anti-inflammatory, analgesic, molluscidal, anti-atherogenic, antioxidant and hepato-protective activities, which have been attributed to some of its phytochemical compositions [22,23].
Another possible mechanism through which the powdered seed sample of G. kola may have caused the lipid lowering effect may be by binding to bile acid in intestine, which will impede their reabsorption. This will subsequently deplete the bile acid pool leading to up regulation of cholesterol 7-α-hydroxylase and increased conversion of cholesterol to bile acids. This causes an increased demand for cholesterol in the liver cells, resulting in the dual effect of increasing transcription and activity of the cholesterol biosynthetic enzyme, HMG-CoA reductase and increase in the number of hepatic LDL receptors. These compensatory effects result in increased clearance of LDL-C from the blood, resulting in decreased serum LDL-C levels. Serum TG levels may increase or remain unchanged [24]. According to Evans and Trease [24], plants may grow well in different situations but fails to produce the same constituents. Plant growth and development, and often the nature and quantity of secondary metabolites are affected by temperature, rainfall, length of day (quantity of light) and altitude. Light determines the amount of glycosides or alkaloids present in a plant. Also, continuous rain can lead to a loss of water-soluble substances from leaves and roots by leaching; this is known to apply to some plants producing alkaloids, glycosides and even volatile oils [24].
4.0 Conclusion
In conclusion, this research work has revealed that G. kola seed has a hypolipidemic effect when consumed and that consumption of G. kola seed may help in the reduction of the incidence of cardiovascular diseases in man and animals and further research should be carried out to determine the tolerable level of G. kolaor a dose that is not lethal and mechanism behind its hypolipidemic effect.
Author’s Contributions
Author FBO is a Veterinary Doctor and a Principal researcher with Forestry Research Institute of Nigeria, he organized the research work with the co-authors and jointly carried out the research work. Author BKO renowned industrial Chemist and also a researcher with the Forestry Research Institute of Nigeria designed and performed the proximate analysis and chemical and biochemical analysis of the experimental diets and samples, while author BOO renowned statistician and also a researcher with the Forestry Research Institute of Nigeria, designed and work on the statistical Analysis of the data obtained and also contributed to the results and in the discussion of the results.
Acknowledgement: Nill
Conflict of Interest: The author declared that no conflict of interest exists
References
1. Alli, S.Y.R and Adanlawo, I.G. (2013). Tissue lipid profile of rats administered saponin extract from the root of bitter kola. Advances in Biochemistry.; 1(1):1-4.
2. Onyeneke, E.C., Adebisi, K.E., Eriyamremu, G.E., Ojeaburu, S.I., Asagba, S.O and Oluba, O.M. (2007). Effect of lipid-based diet on some lipid-metabolizing enzymes.Journal of Medical Sciences.; 7(8):1283-1289.
3. Oluba, O.M., Adeyemi, O., Ojieh, G.C., Adebisi, K.E., Isiosio, I.O and Aboluwoye, C.O. (2008). Effect of dietary cholesterol on some serum enzymes. Journal of Medical Science; 8(4):390-394.
4. Knapp, H.H., Schrott, H., Ma, P., Knopp, R., Chin, B and Gaziano, J.M. (2001). Efficacy and safety of combination simvastatin and colesevelam in patients with primary hypercholesterolemia. The American Journal of medicine; 110:352-360.
5. Omeh, Y.N., Onoja, S.O., Ezeja, M.I., Uchendu, W.C., Okorie, E and Raymond, M. (2014). Quantitative Phytochemical, Proximate Analysis and Hypolipidemic effect of Garcinia kola. British Journal of Medicine & Medical Research 4(36): 5770-5778, 2014. ISSN: 2231-0614
6. Madubunyi, I.I. (2010) Antihepatotoxic principles of Garcinia kola seeds. Clinical Pathology; 21:481- 485.
7. Farombi, E.O., Akanni, O.O and Emerole, G.O. (2002). Antioxidant and scavenging activities of flavonoid extract (kolaviron) of Garcinia kola seeds in vitro. Pharm. Biol.; 91:129– 134.
8. Okunji, C.O., Ware, T.A., Hicks, R.P., Iwu, M.M and Skanchy, (2002) D.J. Capillary electrophoresis determination of biflavanones from Garcinia kola in three traditional African medicinal formulations. PlantaMedicina.; 68:440–444.
9. Eleyinmi, A.F., Bressler, D.C., Amoo, I.A., Sporns, P and Oshodi, A.A. (2006) Chemical composition of bitter cola (Garcinia kola) seed and hulls. Poultry Journal of Food Nutrition.Science. 15(4):395–400.
10. Ward, J.W and Elsea, J.R. (1997). Animal case and use in drug fate and metabolism. In: Edward RG, JeanLH (eds) Methods and techniques, 1st edn., New York: Markel Dekker.
11. Stein, E.A. Lipids, (1987). Lipoproteins and apolipoproteins. In: Tietz, N. W. (Ed). Fundamentals of Clinical Chemistry. 3rdEdn., Philadelphia: W . B Saunders.
12. Tietz, N.W. (1990). Clinical Guide to Laboratory Test, Second Edition, Philadelphia: W.B. Saunders Company.
13. Wacnic, R.G and Alber, J.J. (1978) A comprehensive evaluation of the heparin manganese precipitation Procedure for estimating high density lipoprotein cholesterol. Journal of Lipid Resources. 19:65-76
14. Rifai, N., Warmick, G.R and Remaley, A.T. (2008). Lipids, lipoproteins, apolipoproteins and other cardiovascular risk factors. Pages 402 – 430. In: Burtis CA, Ashwood ER, Bruns DE. (Eds.), Tietz Fundamentals of of Clinical Chemistry, 6th ed. Missouri: Saunders Elsevier.
15. Friedewald, V.T., Levy, R.I and Fredrickson, D.S. (1972). Estimation of low-density lipoprotein cholesterol in plasma, without use of preparative centrifuge. Clinical Chemistry; 18:499502.
16. Krishnaiah, D., Devi, T., Bano, A and Sarbatly, R. (2009). Studies on phytochemical constituents of six Malaysian medicinal plants.Journal of Medicinal Plants Resources 3(2):67-72.
17. Edeoga, H.O., Okwu, D.E and Mbaebie, B.O (2005). Phytochemical constituents of some Nigerian medicinal plants. African Journal of Biotechnology. 4(7):685-688.
18. Association of Official Analytical Chemist. (1990) Official Methods of Analysis, 15th Ed., Arlington, VA.
19. Adesuyi, A.O., Elumm, I.K., Adaramola, F.B and Nwokocha, A.G.M. (2012). Nutritional and Phytochemical Screening of Garciniakola.Advance Journal of Food Science and Technology. 4(1):9-14.
20. Rang, H.P., Dale, M.M., Ritter, J.M and Moore, R.K. (2007). Pharmacology 6th edition, Edinburgh: Churchill livingstone; 2007.
21. Iwu, M.M., Igboko, A.O., Okunji, C.O and Tempesta, M.S. (1990) Antidiabetic and aldose reductase activities of biflavanones of Garcinia kola. J. Pharm. Pharmacol.1990; 42:290-292.
22. Braide, V.P (1991). Antihepatotoxic Biochemical effects of kola viron, a Biflavonoid of Garcinia kola seeds. Phytother. Res. 5:35-37.
23. Fonseca, V.A., Rosenstock, J., Wang, A.C., Truitt, K.E and Jones, M.R. (2008). Colesevelam HCl improves glycemic control and reduces LDL cholesterol in patients with inadequately controlled Type 2 diabetes on sulfonylurea-based therapy.Diabetes.Care. 31(8):1479–1484.
24. Evans, W.C. Trease and Evans Pharmacognosy. (2009)16th ed. Edinburgh: Saunders Elsevier.16:87-89